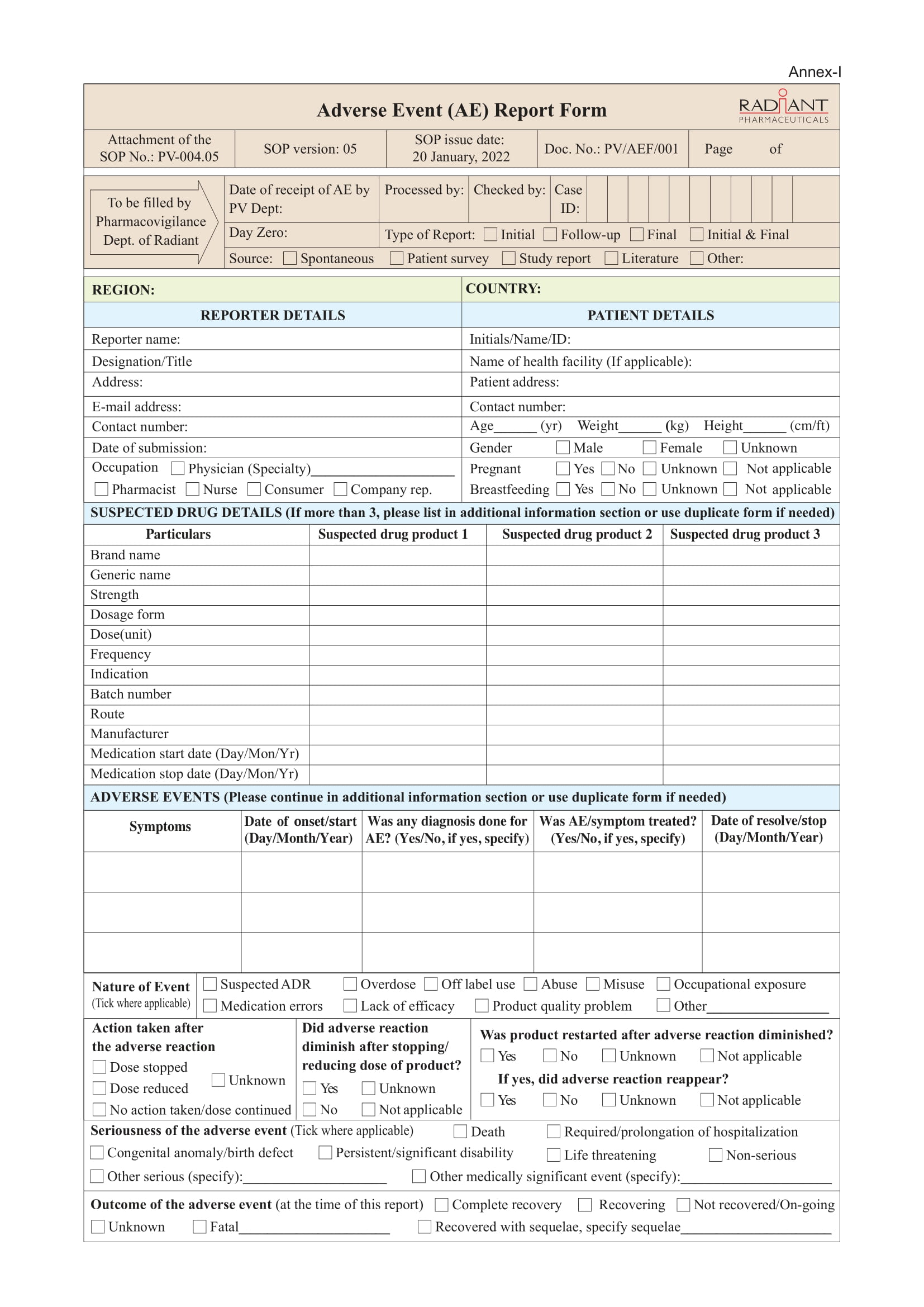
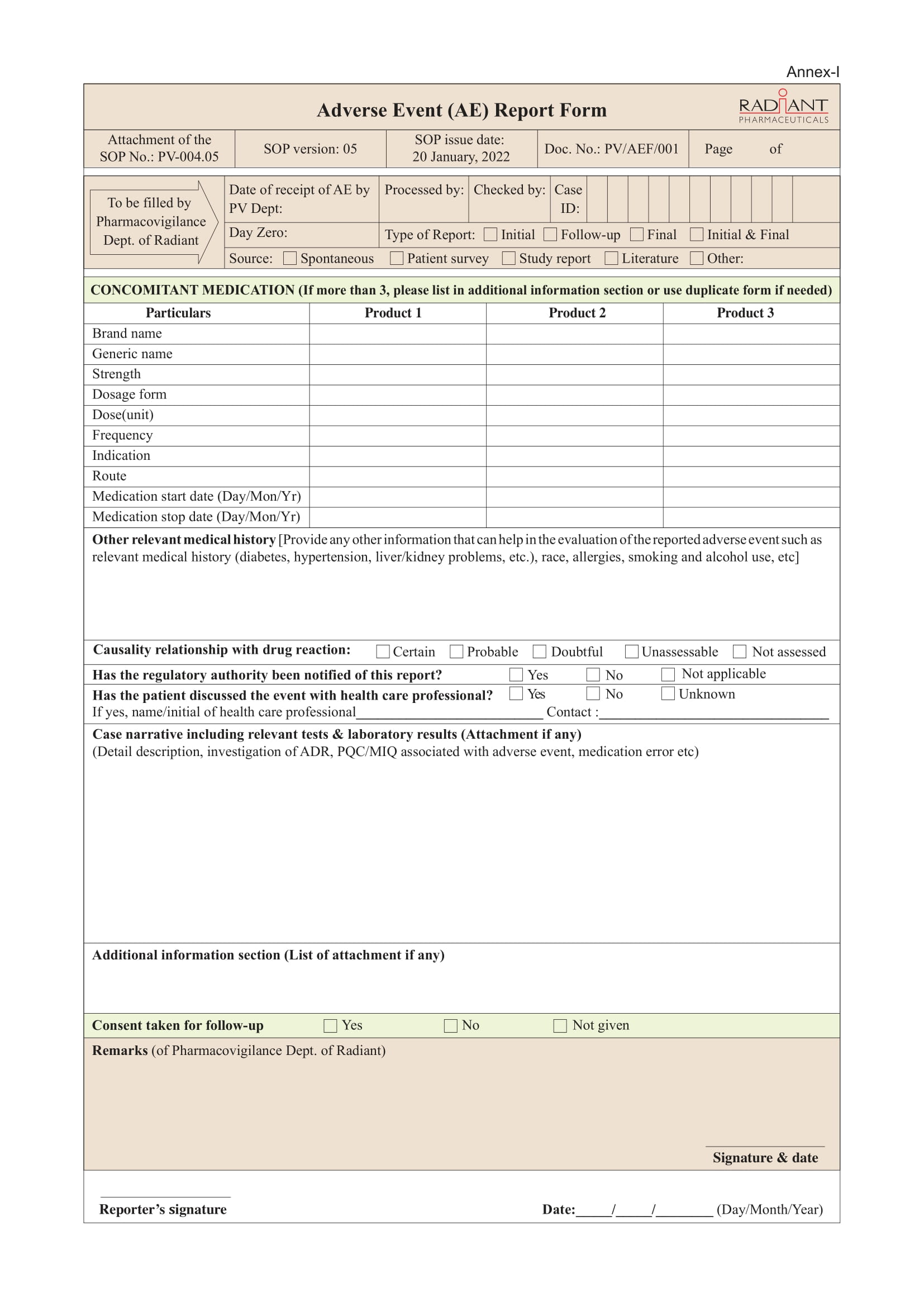
Any adverse event regarding Radiant’s Products (manufactured, imported and/or distributed) can be reported to the Department of Pharmacovigilance.
.png)
pv@radiant.com.bd
.png)
Contact Number
+88 01844018052

Postal/Courier Address
Department of Pharmacovigilance
Radiant Pharmaceuticals Limited
SKS Tower, 7 VIP Road, Mohakhali,
Dhaka 1206, Bangladesh.
All consumers or patients are advised to contact their doctor for advice on medicines or any health problems/ medical emergencies. Pharmacovigilance enhance safer use of medicine by monitoring the safety and efficacy of medicines and by providing reliable, balanced information for the effective assessment of the risk-benefit profile of medicines.
Confidentiality Note: Any information related to the identities of the patient and reporter will be kept confidential unless it is subject to disclose due to regulatory obligation.